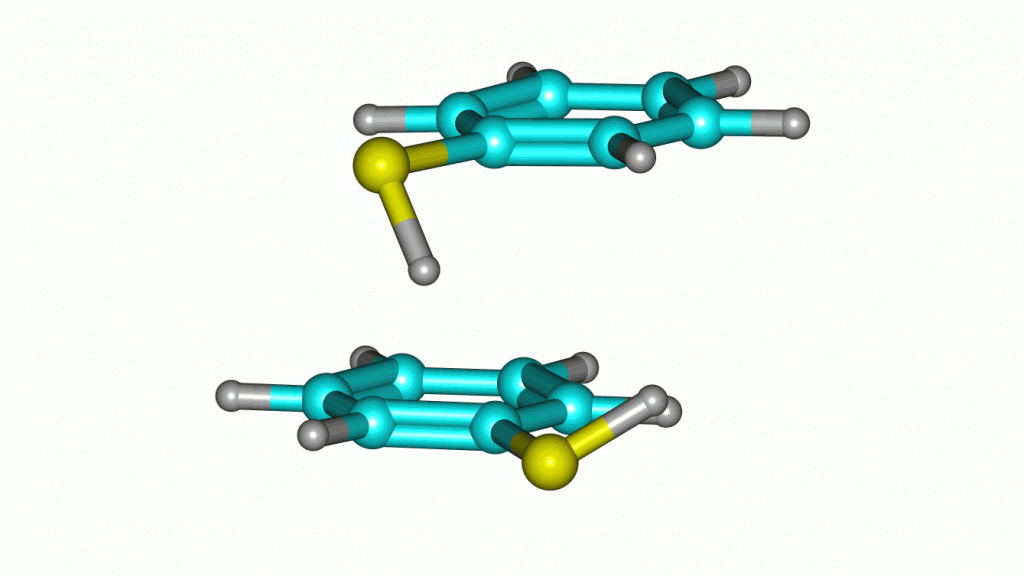
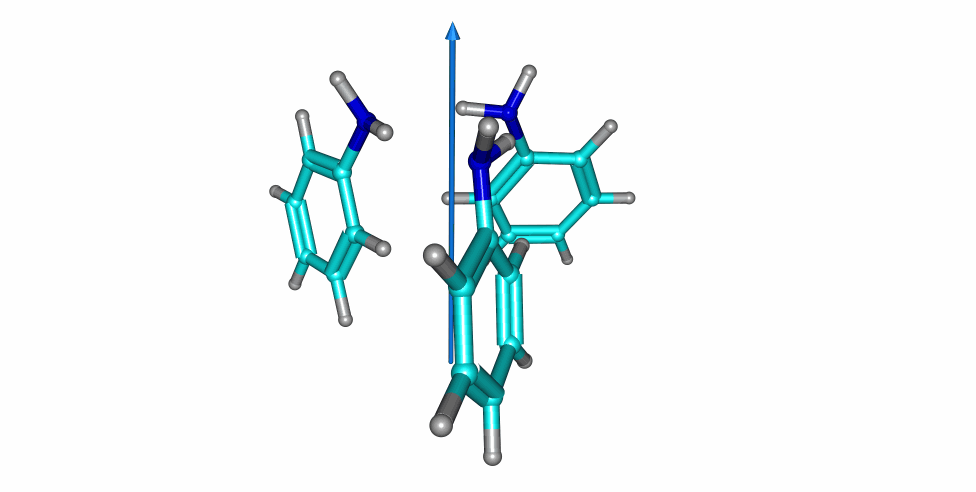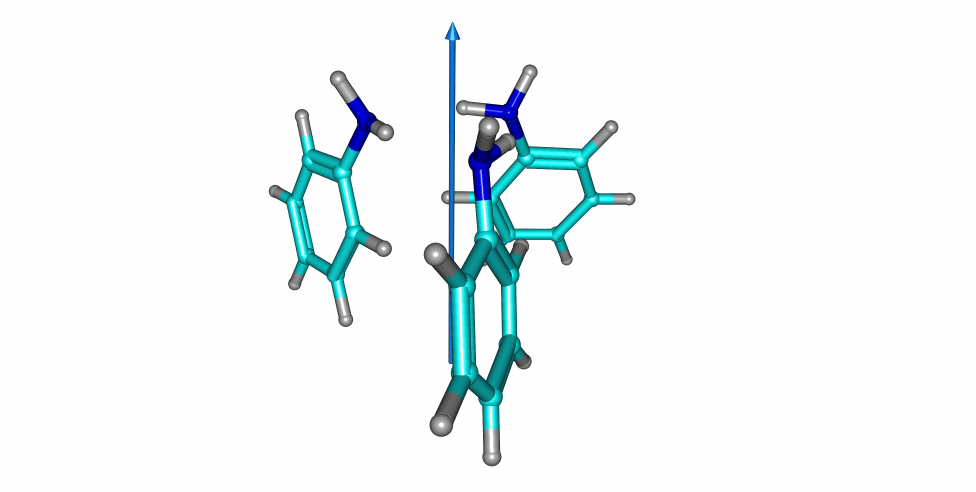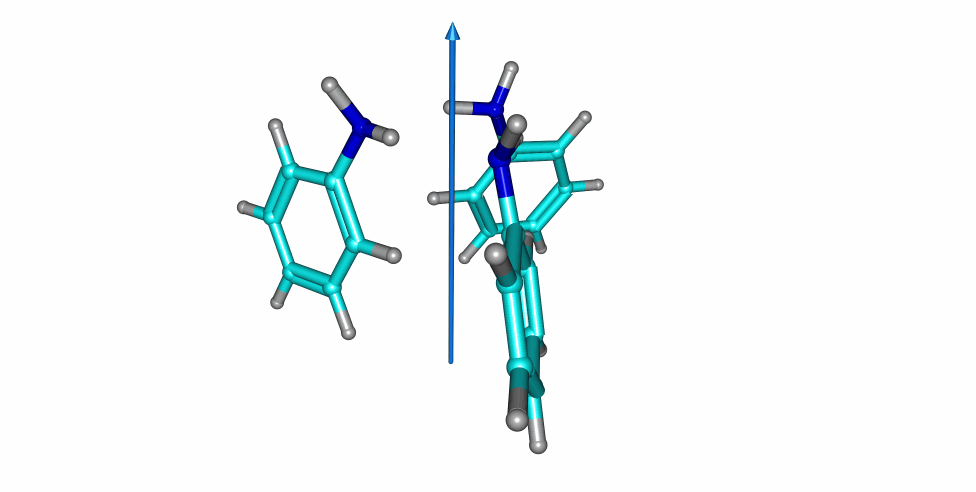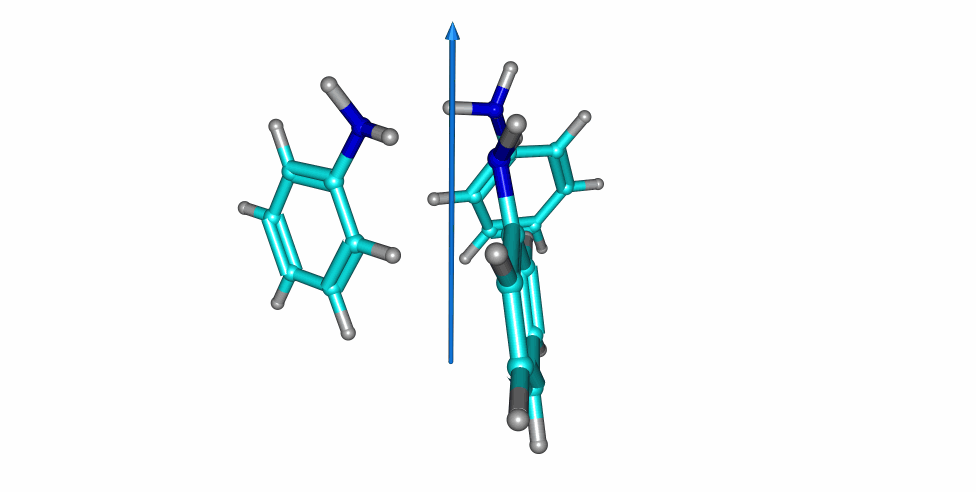
Thiophenol stacking motions
An investigation on the thiophenol dimer and trimer was published in the Journal of Physical Chemistry Letters. The weakly-bound dimer adopts a stacked structure, while simultaneously maintaining a weak S-H···S hydrogen bond. Two isomers were detected for the dimer in the gas-phase. Notoriously, a large-amplitude motion was observed in one of the dimers, exchanging the proton donor and aceptor thiol groups.



You must be logged in to post a comment.